 Atomic # increases by 1, so it has 1 more valence electron than the previous family.The table works because it is based on the structure of atoms, especially the valence e-s (outermost shell).Across a row or down a column the elements’ properties change in a predictable way.An element’s properties can be predicted from its location on the table.Elements are now arranged by increasing atomic number.Published his table in 1869 the newelements were discovered within 16 years.He even predicted the properties of new elements.Mendeleev predicted that the blank spaces would be filled by elements that had not yet been discovered.So he moved the cards into the group it best fit. However, this did not always produce similar groups.Patterns appeared when elements were arranged in order of increasing atomic mass.He listed the elements’ known properties.To help him find a pattern he put all of the known information on individual cards.discovered a system for organizing all of the known elements.1860’s a Russian Scientist named Dmitri Mendeleev.Arranged in rows called periods and columns called groups or families.A way of organizing & classifying elements.29 - Complete fill in the blanks portion of blue packet and turn in. 5 and 6 -Complete yellow Birdley Background: Mendeleev’s table and paste onto p. 28 Periodic Table Trends Notes IQ: On the PT, most elements are non-metals, on left hand side metals, on right hand side non-metals on right hand side metals on left hand side. Modern Periodic Table Periodic – means regular, repeated pattern.Ģ/21 p. OQ: Find the only 2 liquid elements on the PT. The only ones known to create a magnetic field.First elements in groups 8,9, 10 called the iron triad.In space, vast quantities interact with starlight to create spectacular sights such as the Eagle Nebula (seen by the Hubble Space Telescope). Color it GRAY By weight, 75% of the visible universe is hydrogen, a colorless gas.Use Roman numerals I thru VIII to show the families.A Flash animation by Mike Stanfill, Private Hand IQ: What are some ways for organizing information in Science? Why do we organize information?.Laminate the fill-in-the-blank worksheets (or put them in plastic page protectors) and use dry erase markers to fill in the answers.Each of you needs1 of each of the following colors: Red Orange Blue Green Purple Pink Brown.At first, you may want to use the alphabetical list of the elements so the students know how to spell each word. They'll need to write down the atomic number and then the full element name next to it. For the worksheets with the element names missing, have your students use a separate piece of paper to write their answers.For nonmetals, students should circle the entire nonmetals group since some of the elements in the group also belong to halogens and noble gases.Students can color-code any of the black and white charts. There's a color key at the bottom of each page they can fill in. They'll be coloring the different classifications of elements, such as halogens, transition metals, etc. Have your student use colored pencils to color-code the black and white version of the full chart.Download the Periodic Table of the Elements worksheet set HERE Some Tips for Using the Worksheets You're welcome to download the Periodic Table of the Elements set of worksheets for your personal use. Half the worksheets cover all the known elements (for older students), but the other half cover only the 40 most common elements (for younger or beginning chemistry students). I've also made the worksheets for two different learning levels. And there's even a version that has no atomic number, symbol, or name - the student has to fill out everything! Some of the worksheets have the element names removed, or the chemical symbols, or both. The other 10 pages are different versions of the chart in black and white that allow the student to learn the element names, chemical symbols, and atomic numbers in different ways. The set of worksheets includes a printable, color-coded periodic table of the elements and an alphabetical list of the elements. And as usual, I couldn't find exactly what I wanted online, so I made up my own and I'm making it available to my readers as a free download. 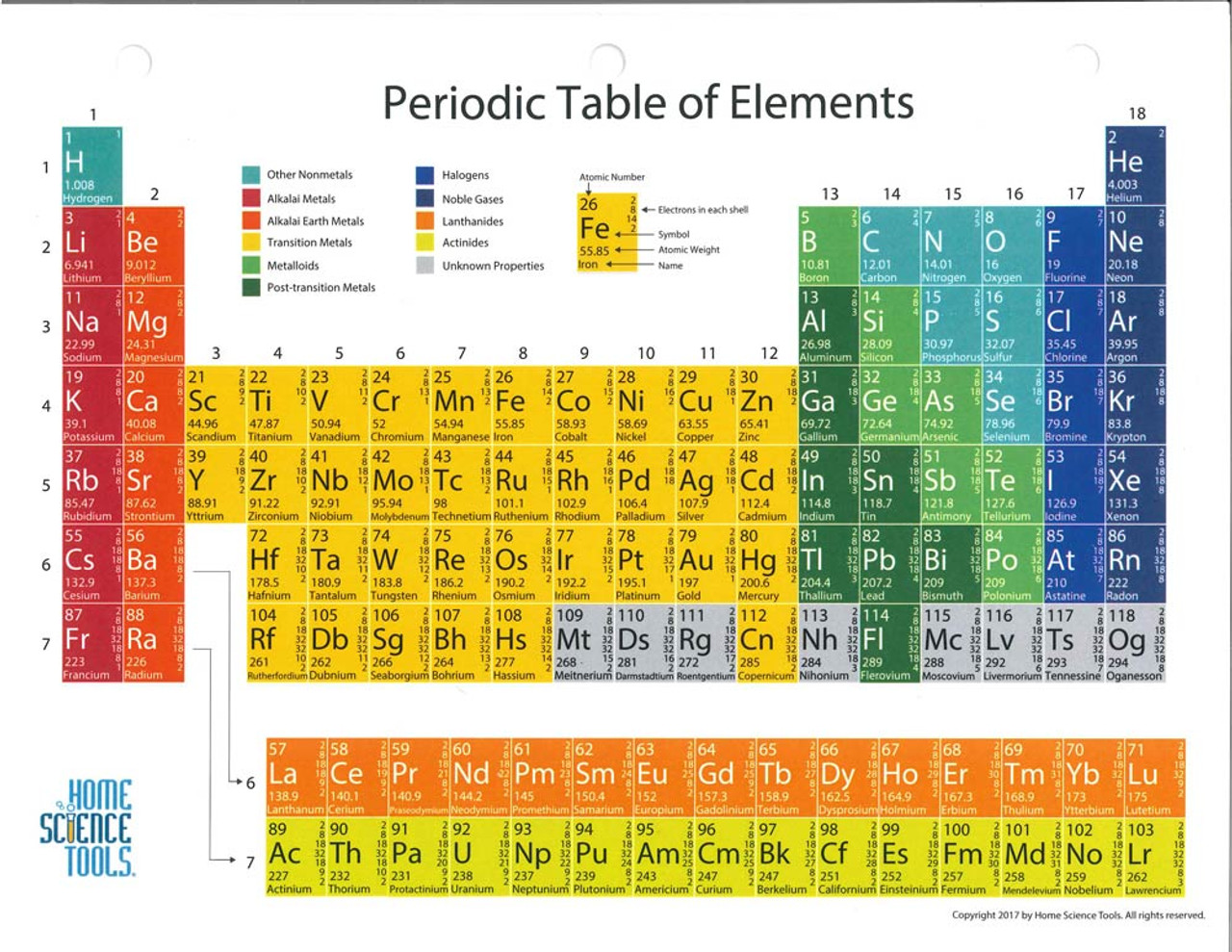
So I've come up with a different method for her, hopefully one that will be even more effective. However, I'm not liking the flashcard method of learning the element names. We're liking it so far, and she's learning alot. Joely decided she wanted to learn about chemistry this year, so I did some research and purchased Christian Kids Explore Chemistry. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
